Learn the basics of CAR T-cell therapy and how it is being used in the SJCAR19 clinical trial at St. Jude

Aimee Talleur, MD, explains the basics of CAR T-cell therapy and how it is used in the SJCAR19 clinical trial at St. Jude.
CAR T-cell therapy has generated excitement throughout the medical community as a treatment for many types of cancer. But what is this treatment and how does it work?
Your immune system is really powerful. It is constantly surveying your body, looking for intruders. When the immune system sees something that looks different, it says, “You don’t belong here.” It then attacks the interloper and clears it from the body. This is what happens with infections all of the time.
When you get cancer, your immune system, for whatever reason, wasn’t able to recognize the cancer cell as different and therefore could not kill it. CAR T-cell therapy seeks to help the immune system find tumor cells more efficiently, while still using the immune system’s power to destroy the abnormal cells.
What is a CAR?
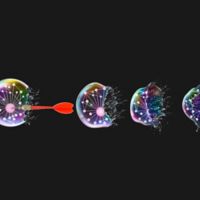
CAR stands for chimeric antigen receptor. This receptor is designed to target a specific molecule that is present on the surface of a cancer cell.
In CAR T-cell therapy, doctors first remove T cells from a person’s blood. The immune cells are then engineered in a lab so that each T cell has a CAR on its surface, making it into a CAR T cell. Once the transformed CAR T cells are infused into the patient, the cells roam around the body looking for their specific target. (In our SJCAR19 clinical trial, that target is the CD19 molecule.) When they find the intended target, the engineered immune cells bind to the cancer cell and kill it.
How the process unfolds
- The first step is to ensure that a patient has a type of tumor for which CAR T-cell therapy is available. Because this therapy is specific and looks for a certain marker on a tumor cell, it’s unlike chemotherapy, which kills cells in a more general fashion. CAR T-cell therapy targets particular tumor cells.
- Next comes collection of the T cells. It takes a few days to do testing and figure out if the patient or donor is a good candidate for collection. The collection, called leukapheresis or apheresis, typically lasts four to eight hours.
- The T cells go to the manufacturing facility (at St. Jude, that’s the Children’s GMP, LLC). There, the T cells are transformed into CAR-T cells. That process takes about 10 to 14 days, and then the engineered cells are tested to ensure they meet stringent quality-assurance standards.
- Meanwhile, the patient is evaluated. We make sure patients are as healthy as possible so that they can better withstand potential side effects of this treatment. This evaluation takes about a week or so. After the evaluation is completed and the patient is deemed ready to get the CAR T cells, the individual is typically admitted to the hospital to begin treatment. There, several days of chemotherapy may be given to prepare the child’s body for receiving the CAR-T cells.
- The infusion of the cells is quick, lasting only a few minutes. The patient remains in the hospital for a while, waiting for the cells to work.
- Once the cells are infused, they begin to grow. It’s different than chemotherapy, because this is a living cell product. The CAR-T cells roam around the body looking for their target. When they see the target, they multiply so that they have a bigger army to fight the cancer.
Possible side effects of CAR T-cell therapy
- Inflammation. When you get sick, you may have fevers, aches and pains. It’s all caused by inflammation from an active immune system. Similarly, when the CAR-T cells become active, they also can cause inflammation. Some of the main side effects from CAR T-cell therapy are related to the cells doing what we want them to do. But patients also have the potential to get sick from this inflammation. That’s one of the reasons we look at patients head to toe beforehand, to make sure they’re as healthy as they can be. We watch patients closely to ensure they can withstand that inflammation.
- Cytokine release syndrome, or CRS, is a general term for the inflammation. As your immune cells get active and divide, they make things called cytokines. These chemical signals help promote CAR T-cell growth and their ability to kill cancer cells. That process also creates inflammation. So patients can get fevers, blood pressure variations, breathing issues, neurologic changes, or really a change to the function of any organ. These side effects can be mild or severe.
- Tumor lysis syndrome. The death of the tumor can also cause side effects. This is called tumor lysis syndrome. Suppose you have a lot of tumor cells. As the cells die, they release electrolytes and other materials into your bloodstream, which can cause problems with your kidneys or other organs.
- Off-tumor effects. We also have to watch for what we call on-target, off-tumor effects. The CAR-T cells look for a marker on the surface of a tumor cell, but that marker may also be present in some normal cells in the body. We, as researchers, need to know ahead of time what normal things in your body may also have that surface marker so we can manage these problems.
Advantages of CAR T-cell therapy
If you think about how chemotherapy, radiation and other non-immune therapies work, they’re nonspecific. It’s like throwing a big bucket of water at everything as opposed to going for the source of the fire. It’s amazing that you can use the body’s own immune system and make it work better to fight cancer.
CAR T-cell therapy is quite powerful. For patients with acute lymphoblastic leukemia (ALL) in particular, CAR T-cell therapy is able to get the vast majority of patients into remission, sometimes for the first time since they began treatment. The length of remission varies between patients, and we do not know enough yet to determine up front for whom CAR-T therapy can be their final therapy. But, getting someone into even a temporary remission may provide a patient with the best chance to receive other therapies, such as hematopoietic cell transplant, which we know has a high chance at providing a long-term cure.
There are certain patient populations whose type of cancer simply does not respond well to chemotherapy. As we learn more and more about CAR T-cell therapy, it might be a frontline option for these patients. But we’re not there yet. As a field, that’s definitely something we are pursuing.
SJCAR19
The St. Jude clinical trial SJCAR19 is designed for children, adolescents and young adults with ALL whose cancer has never responded to treatment, or has returned after treatment. This study targets the CD19 molecule on leukemia cells.
SJCAR19 features modified T cells that are similar to an FDA-approved product already on the market. The way the CAR finds CD19 is the same and the way it stimulates the T cells is the same, but we’ve made some changes to the vector backbone that will hopefully make it safer in the long run. Also, changes we’ve made to the manufacturing process may make it a better product that can last longer in the body or be more effective in killing cancer cells. These are things we will evaluate as part of the clinical trial.
St. Jude is a wonderful place where we provide state-of-the-art care for patients. We have a lot of resources available and a strong multidisciplinary team. There are many people involved in the care of every single patient, and we do everything we can to provide the best care to each child who comes through our doors.
SJCAR19 is the first of many CAR-T trials we’re planning. Eventually, we hope to use CAR T-cell therapy to treat acute myeloid leukemia, as well as some solid tumors and brain tumors. For me, this is not a job. It’s my ideal calling: to be part of a family’s journey, and also to be part of incredible science that’s growing and changing.