Lando: Star Wars smuggler or possible ally against Alzheimer’s disease?

Gambler, con artist, mining engineer, Cloud City administrator and LC3-associated endocytosis acronym, LANDO is now the name given to a discovery from the lab of Doug Green, PhD.
The story starts not a longtime ago in a galaxy far, far away, but in 2018 on the patio of a Pinch District pub in Memphis.
Rather than humans and space aliens in a high-stakes card game, the happy hour crowd that spring evening included Doug Green, PhD, chair of the St. Jude Department of Immunology, and the scientists and staff from his laboratory. The movie “Solo: A Star Wars Story” had just been released and the talk that night was of Star Wars and science.
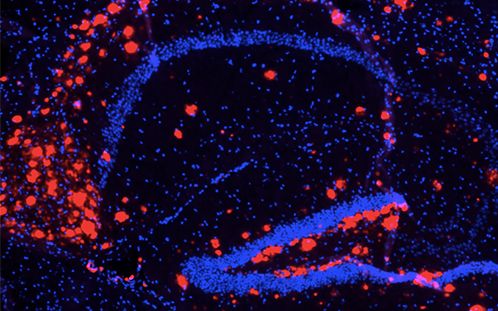
The image of red-stained β-amyloid protein and blue-stained cell nuclei all on a black background really looked like something from Star Wars to Brad Heckmann, PhD, a postdoctoral fellow at St. Jude.
Manipulating the immune response
The scientific discussion included talk of a novel process Brad Heckmann, PhD, a postdoctoral fellow in Green’s lab, had identified while exploring ways to modulate the immune response in the brain. Learning to manipulate the immune response could lead to new treatments for brain tumors and neurodegenerative disorders like Alzheimer’s disease that are characterized by chronic inflammation.
Heckmann was studying microglial cells, which are the primary immune cells in the brain. The cells regulate not only the immune response, but also play a role in brain development and function.
The LC3 protein and autophagy
In his research, Heckmann was investigating whether proteins involved in autophagy, which translates into self-eating, might also play a role in modulating the immune response. Cells use autophagy to recycle nutrients and clear damaged or unneeded cells and cell parts. For this study, researchers used a mouse model of Alzheimer’s that models disease inflammation in the brain. The scientists knew the toxic β-amyloid proteins associated with Alzheimer’s were taken up and cleared from the brain by microglial cells.
By the time the Green lab gathered that evening, researchers had some more answers. They had evidence that several of the autophagy proteins, including the LC3 protein, played a key role in the microglial cell endocytosis that prevented the build-up of β-amyloid proteins in mice.
Around the lab the newly identified process was known as LC3-associated endocytosis — not exactly a name that rolled off the tongue. In biology, discovery typically includes naming rights. Green’s lab wanted a name or nickname that was catchy and relatable. A name that like Star Wars was likely to endure.
LANDO at work
As happy hour progressed and talk ping-ponged between science and Star Wars, Green mentioned Lando Carlissian, the charming smuggler and pilot in the first Star Wars movie who returned in the latest movie. “At some point it just clicked in Doug’s mind and LC3-associated endocytosis became LANDO,” Heckmann said, explaining the origins of the acronym.
What sold Heckmann on the acronym were the images of LANDO at work in microglial cells. The images included β-amyloid protein stained red. The toxic protein accumulated in the brain when LANDO was disabled. “The image of red-stained β-amyloid protein and microglial cells stained green all on a black background really looked like something from Star Wars,” he said.
LANDO recently made its scientific debut in the journal Cell.